Pharmacological prediction of Marchantia polymorpha: GC-MS and molecular docking approaches
DOI:
https://doi.org/10.23917/bioeksperimen.v12i1.15983Keywords:
Marchantia polymorpha, GC-MS, Bioactive compound, Molecular docking, SwissADME, Pass OnlineAbstract
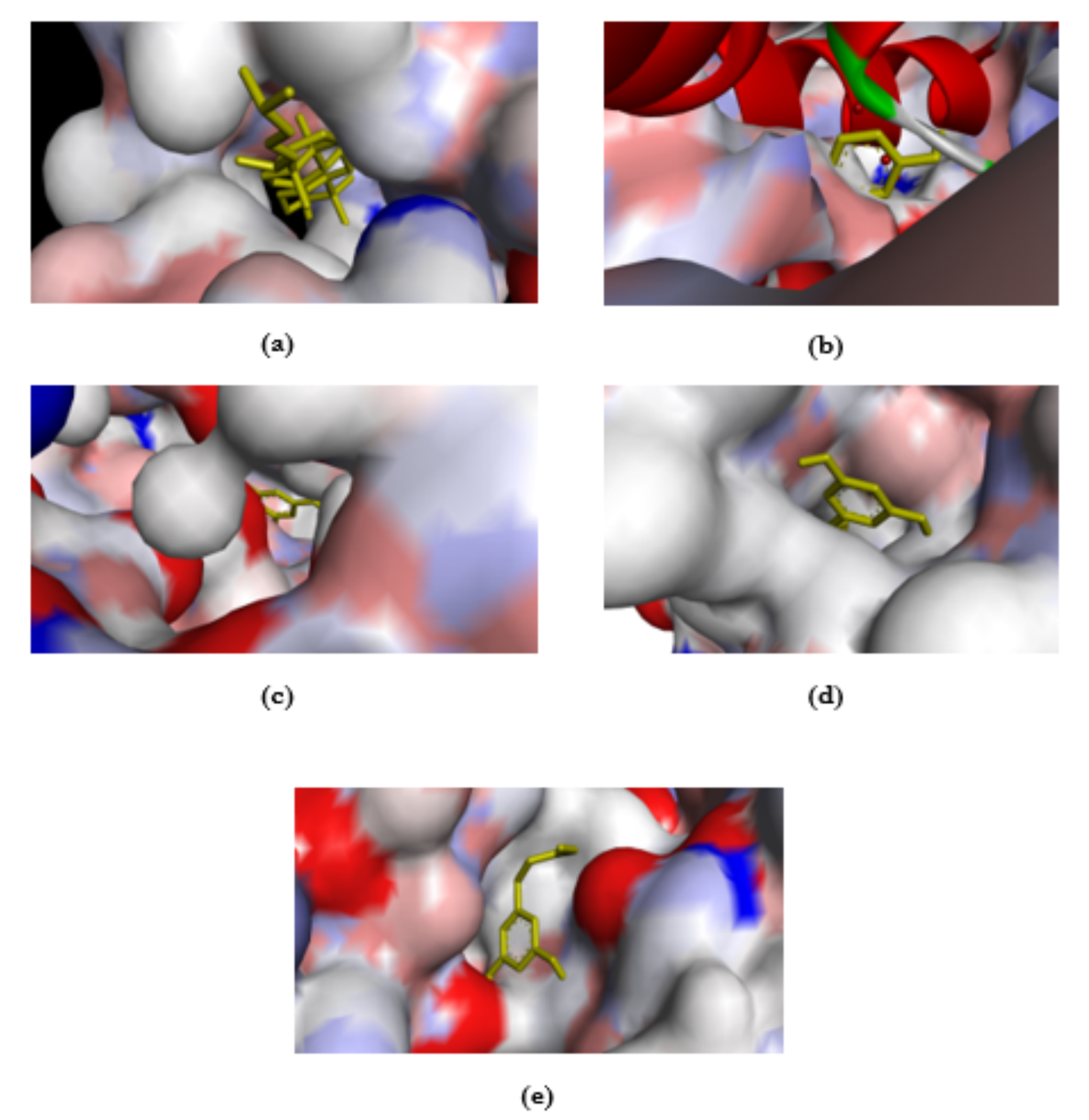
Marchantia polymorpha is the liverworts that can be found abundantly in the Mount Pasang Jember area. The metabolite profiling of M. polymorpha has not yet been conducted. The method used in this study was untargeted metabolite profiling using GC-MS, and the results of the metabolite compound profiling were analyzed in silico using bioinformatics-based, namely PASS Online, SwisADME, and Molecular Docking using PyRx 8.0 with AutodockVina. The aims of docking with this protein was adjusted to the PASS Online results, namely alkenylglycerophosphocholine hydrolase inhibitor and antiseborrheic, which are closely related to antifungals. The natural metabolite compounds detected from M. polymorpha were fatty acids (20%), terpenoids (16%), and phenolics (10%). Terpenoids-phenolics compounds were analyzed in silico to predict pharmacological potential. Terpenoids showed that the compound with the highest Pa value was 3,7-Cyclodecadiene (Pa=0.920), predicted to be an alkenylglycerophosphocholine hydrolase inhibitor, while the phenolic compound with the highest Pa value was hydroquinone (Pa=0.927), predicted to be an antiseborrheic. Based on SwissADME in silico Druglikeness, five compounds out of a total of six terpenoid and phenolic compounds showed compliance with Lipinski's theory. In addition, the in silico results also showed a bioavailability score of 0.55. Molecular docking was performed on the target protein sterol 14-α-demethylase (CYP51) from Candida albicans (PDB ID: 5TZ1). This protein is representative of antifungal agents. The results of molecular docking showed that the compound 3,7-Cyclodecadiene consistently had the strongest binding affinity value of -10.1 kcal/mol with residues ILE A:55, ALA A:62, PHE A:58, and TRP B:5 These results imply that further research on M. polymorpha metabolites should be conducted using comprehensive methods to explore their potential in the field of health.
Downloads
References
Agu, P. C., Afiukwa, C. A., Orji, O. U., Ezeh, E. M., Ofoke, I. H., Ogbu, C. O., Ugwuja, E. I., & Aja, P. M. (2023). Molecular docking as a tool for the discovery of molecular targets of nutraceuticals in diseases management. Scientific Reports, 13(1), 1–18. https://doi.org/10.1038/s41598-023-40160-2
Ahmmed, F., Islam, A. U., Mukhrish, Y. E., Bakri, Y. El, Ahmad, S., Ozeki, Y., & Kawsar, S. M. A. (2023). Efficient Antibacterial/Antifungal Activities: Synthesis, Molecular Docking, Molecular Dynamics, Pharmacokinetic, and Binding Free Energy of Galactopyranoside Derivatives. Molecules, 28. https://doi.org/https://doi.org/10.3390/molecules28010219
Antypenko, L., Antypenko, O., Karnaukh, I., Rebets, O., Kovalenko, S., & Arisawa, M. (2023). Synthesis, Antimicrobial Activity, Molecular Docking, and Perspectives. Arch Pharm Chemistry in Life Science, January, 1–15. https://doi.org/10.1002/ardp.202300029
Arata, C., Misztal, P. K., Tian, Y., Lunderberg, D. M., Kristensen, K., Novoselac, A., Vance, M. E., Farmer, D. K., Nazaroff, W. W., & Goldstein, A. H. (2021). Volatile organic compound emissions during HOMEChem. In Indoor Air. 31(6). https://doi.org/10.1111/ina.12906
Ayodele, P. F., Bamigbade, A. T., Bamigbade, O. O., Adeniyi, I. A., Tachin, E. S., Seweje, A. J., & Farohunbi, S. T. (2023). Illustrated Procedure to Perform Molecular Docking Using PyRx and Biovia Discovery Studio Visualizer : A Case Study of 10kt With Atropine. HH Publisher : Progress in Drug Discovery and Biomedical Science, 6, 1–31. https://doi.org/10.36877/pddbs.a0000424
Baroroh, U., Muscifa, Z. S., Destiarani, W., Rohmatulloh, F. G., & Yusuf, M. (2023). Molecular Interaction Analysis and Visualization of Protein-Ligand Docking using Biovia Discovery Studio Visualizer. Indonesian Journal of Computational Biology, 2(1), 22–30. https://doi.org/10.24198/ijcb.v2i1.46322
Bozelli, J. C., Azher, S., & Epand, R. M. (2021). Plasmalogens and Chronic Inflammatory Diseases. Frontiers in Physiology, 12(October), 1–19. https://doi.org/10.3389/fphys.2021.730829
Cai, Y. Y., Chen, T., & Cao, J. F. (2022). Antimicrobial and Antioxidant Metabolites From the Cultured Suspension Cells of Marchantia polymorpha L. Natural Product Communications, 17(4), 1–7. https://doi.org/10.1177/1934578X221096172
Chikowe, I., Bwaila, K. D., Ugbaja, S. C., & Abouzied, A. S. (2024). GC–MS Analysis, Molecular Docking, and Pharmacokinetic Studies af Multidentia crassa Extracts’ Compounds for Analgesic and Anti-Inflammatory Activities in Dentistry. Scientific Reports, 14(1), 1–18. https://doi.org/10.1038/s41598-023-47737-x
Daina, A., Michielin, O., & Zoete, V. (2017). SwissADME: A Free Web Tool To Evaluate Pharmacokinetics, Drug-Likeness and Medicinal Chemistry Friendliness of Small Molecules. Scientific Reports, 7(1), 1–13. https://doi.org/10.1038/srep42717
Daina, A., & Zoete, V. (2019). Application Of The Swissdrug design Online Resources in Virtual Screening. International Journal of Molecular Sciences, 20(18). https://doi.org/10.3390/ijms20184612
Desai, T. H., & Joshi, S. V. (2019). In silico Evaluation of Apoptogenic Potential and Toxicological Profile of Triterpenoids. Indian Journal of Pharmacology, 49(5), 344–347. https://doi.org/10.4103/ijp.IJP
Deshmukh, H. S., Adole, V. A., & Jagdale, B. S. (2025). Design, synthesis, Biological Evaluation, and Computational Insights of 2 ‑( Aryl ) derivatives as potent Antitubercular and Antibacterial Agents. BMC Chemistry. https://doi.org/10.1186/s13065-025-01405-5
Dorninger, F., Werner, E. R., Berger, J., & Watschinger, K. (2022). Regulation of Plasmalogen Metabolism and Traffic in Mammals: The Fog Begins To Lift. Frontiers in Cell and Developmental Biology, 10(August), 1–22. https://doi.org/10.3389/fcell.2022.946393
Druzhilovskiy, D. S., Rudik, A. V., Filimonov, D. A., Gloriozova, T. A., Lagunin, A. A., Dmitriev, A. V., Pogodin, P. V., Dubovskaya, V. I., Ivanov, S. M., Tarasova, O. A., Bezhentsev, V. M., Murtazalieva, K. A., Semin, M. I., Maiorov, I. S., Gaur, A. S., Sastry, G. N., & Poroikov, V. V. (2017). Computational Platform Way2Drug: from the Prediction of Biological Activity to Drug Repurposing. Russian Chemical Bulletin, 66(10), 1832–1841. https://doi.org/10.1007/s11172-017-1954-x
Elsaman, T., Khalid, M., Awadalla, A., & Mohamed, M. S. (2025). Identification of Microbial-Based Natural Products as Potential CYP51 Inhibitors for Eumycetoma Treatment : Insights from Molecular Docking, MM-GBSA Calculations, ADMET Analysis, and Molecular Dynamics Simulations. Pharmaceuticals, 18. https://doi.org/10.3390/ph18040598
Emwas, A. M., Al-talla, Z. A., Yang, Y., & Kharbatia, N. M. (2015). Chapter 8 Gas Chromatography – Mass Spectrometry of Biofluids and Extracts. Metabonomics, 1277, 91–112. https://doi.org/10.1007/978-1-4939-2377-9
Farhan, A. M., Rosida, W., & Ulum, F. B. (2025). In Silico Biological Activity Prediction of Bioactive Compounds from Dumortiera hirsuta (Sw.) Nees. Using Way2Drug PASS Online. Life Science and Biotechnology, 3(1), 13–19.
Farhan, A. M., Setyati, D., Gilani, N. A., Su’udi, M., & Ulum, F. B. (2025). Phytochemistry Profile and Antioxidant Activity of Dumortiera hirsuta (Sw.) Nees from Gumitir, East Java. Hayati Journal of Biosciences, 32(6). https://doi.org/10.4308/hjb.32.6.1643-1653
Farhan, A. M., Zulfikar, M., Masayu, A., Azizah, A. N., Risqi, A. Z., Shodiq, A. H., Shodiq, M. H., Biologi, J., Matematika, F., Alam, P., Jember, U., Hewan, J. K., Jember, U., Fisika, J., Matematika, F., Alam, P., & Jember, U. (2025). Marchantia polymorpha: Studi Profil dan Kandungan Senyawa Bioaktif Potensial sebagai Obat di Masa Mendatang. Jurnal Penelitian Sains, 27, 1–6. https://doi.org/10.56064/jps.v27i2.1101
Fawwaz, M., Mishiro, K., Purwono, B., Nishii, R., & Ogawa, K. (2024). Synthesis and Evaluation of A Rociletinib Analog as Prospective Imaging Double Mutation L858R/T790M in Non-Small Cell Lung Cancer. Journal of Pharmacy and Pharmacognosy Research, 12(2), 231–242. https://doi.org/10.56499/jppres23.1743_12.2.231
Fiehn, O. (2017). Metabolomics by Gas Chromatography-Mass Spectrometry: the Combination of Targeted and Untargeted Profiling. In Curr Protoc Mol Biol, 7(3). https://doi.org/10.1002/0471142727.mb3004s114.
Filatov, V. A., Kulyak, O. Y., & Kalenikova, E. I. (2023a). Chemical Composition and Antimicrobial Potential of a Plant-Based Substance for the Treatment of Seborrheic Dermatitis. Pharmaceuticals, 16(3). https://doi.org/10.3390/ph16030328
Filatov, V. A., Kulyak, O. Y., & Kalenikova, E. I. (2023b). The Development of Medical Shampoo with a Plant-Based Substance for the Treatment of Seborrheic Dermatitis. Medical Sciences Forum, 21. https://doi.org/10.3390/ph16030328
Frolova, N., Orlova, A., Popova, V., & Bilova, T. (2025). Gas Chromatography – Mass Spectrometry (GC-MS) in the Plant Metabolomics Toolbox : Sample Preparation and Instrumental Analysis. Biomolecules, 1–33. https://doi.org/10.3390/biom16010016
Hargrove, T. Y., Friggeri, L., Wawrzak, Z., Qi, A., Hoekstra, W. J., Schotzinger, R. J., York, J. D., Guengerich, F. P., & Lepesheva, G. I. (2017). Structural Analyses of Candida albicans sterol 14 -demethyl- ase complexed with Azole Drugs Address the Molecular Basis of Azole-Mediated Inhibition of Fungal Sterol Biosynthesis. Journal of Biological Chemistry, 292(13), 6728–6743. https://doi.org/10.1074/jbc.M117.778308
Hussein, H. M. (2016). Analysis Of Trace Heavy Metals and Volatile Chemical Compounds of Lepidium sativum using Atomic Absorption Spectroscopy, Gas Chromatography-Mass Spectrometric and Fourier-Transform Infrared Spectroscopy. Research Journal of Pharmaceutical, Biological and Chemical Sciences, 7(4), 2529–2555.
Ilmiah, I. I., Mufidi, F. F., Taruna, E., Perkasa, R., Dzaky, C., Isdarmanto, P., Shodiq, M. H., Shodiq, A. H., & Farhan, A. M. (2025). Chemical Profiling of Locally Grown Zingiber officinale Roscoe (Ginger) Rhizomes using GC-MS and Biological Activities. 3(1), 1–5.
Jadhav, A. K., Khan, P. K., & Karuppayil, S. M. (2020). Phytochemicals As Potential Inhibitors of Lanosterol 14 -Demethylase ( Cyp51 ) Enzyme : An In Silico Study on Sixty Molecules. International Journal of Applied Pharmaceutics, 12(4). http://dx.doi.org/10.22159/ijap.2020.v12s4.40100.
Kajikawa, M., Yamato, K. T., Kanamaru, H., Sakuradani, E., Shimizu, S., Fukuzawa, H., Sakai, Y., & Ohyama, K. (2003). MpFAE3, a β-ketoacyl-coA Synthase Gene in the Liverwort Marchantía polymorpha L., is Preferentially Involved in Elongation of Palmitic Acid to Stearic Acid. Bioscience, Biotechnology and Biochemistry, 67(8), 1667–1674. https://doi.org/10.1271/bbb.67.1667
Kamatou, G. P. P., & Viljoen, A. M. (2017). Comparison of fatty acid methyl esters of Palm and Palmist Oils Determined by GCxGC–ToF–MS and GC–MS/FID. South African Journal of Botany, 112, 483–488. https://doi.org/10.1016/j.sajb.2017.06.032
Kumar, S., Kempinski, C., Zhuang, X., Norris, A., Mafu, S., Zi, J., Bell, S. A., Nybo, S. E., Kinison, S. E., Jiang, Z., Goklany, S., Linscott, K. B., Chen, X., Jia, Q., Brown, S. D., Bowman, J. L., Babbitt, P. C., Peters, R. J., Chen, F., & Chappell, J. (2016). Molecular Diversity of Terpene Synthases in the Liverwort Marchantia polymorpha. Plant Cell, 28(10), 2632–2650. https://doi.org/10.1105/tpc.16.00062
Lemos, M. F., de Andrade Salustriano, N., de Souza Costa, M. M., Lirio, K., da Fonseca, A. F. A., Pacheco, H. P., Endringer, D. C., Fronza, M., & Scherer, R. (2022). Chlorogenic Acid and Caffeine Contents and Anti-Inflammatory and Antioxidant Activities of Green Beans of Conilon and Arabica Coffees Harvested With Different Degrees of Maturation. Journal of Saudi Chemical Society, 26(3). https://doi.org/10.1016/j.jscs.2022.101467
Lohidashan, K., Rajan, M., Ganesh, A., Paul, M., & Jerin, J. (2018). Pass and Swiss ADME Collaborated in Silico Docking Approach to The Synthesis of Certain Pyrazoline Spacer Compounds For Dihydrofolate Reductase Inhibition And Antimalarial Activity. Bangladesh Journal of Pharmacology, 13(1), 23–29. https://doi.org/10.3329/bjp.v13i1.33625
Muslikh, F. A., Suryanto, Nahdhia, N., Susilawati, D., Sari, F., Nugroho, S. A., Werdiningsi, W., & Basuki, D. R. (2025). Prediksi Druglikeness dan Potensi Aktivitas Biologis Senyawa Metabolit Sekunder Daun Asam Jawa (Tamarindus indica). Jurnal Sintesis: Penelitian Sains, Terapan Dan Analisisnya, 6(1), 49–60. https://doi.org/10.56399/jst.v6i1.264
Ninkuu, V., Zhang, L., Yan, J., Fu, Z., Yang, T., & Zeng, H. (2021). Biochemistry of Terpenes and Recent Advances in Plant Protection. International Journal of Molecular Sciences, 22(11). https://doi.org/10.3390/ijms22115710
Nowaczyński, F., Nicoletti, R., Zimowska, B., & Ludwiczuk, A. (2025). Marchantia polymorpha as a Source of Biologically Active Compounds. Molecules, 30(3), 1–19. https://doi.org/10.3390/molecules30030558
Pagadala, N. S., Syed, K., & Tuszynski, J. (2017). Software for Molecular Docking: a review. Biophysical Reviews, 9(2), 91–102. https://doi.org/10.1007/s12551-016-0247-1
Paggi, J. M., Pandit, A., & Dror, R. O. (2024). The Art and Science of Molecular Docking. Annual Review of Biochemistry, 93(1), 389–410. https://doi.org/10.1146/annurev-biochem-030222-120000
Pan, M., Lei, Q., Zang, N., & Zhang, H. (2019). A strategy based on GC-MS/MS, UPLC-MS/MS and Virtual Molecular Docking for Analysis and Prediction of Bioactive Compounds in Eucalyptus globulus Leaves. International Journal of Molecular Sciences, 20(16). https://doi.org/10.3390/ijms20163875
Pannu, A., Kapila, S., Secrain, S., Sabharwal, H., Sethi, M., Sharma, S., & Dogra, N. (2024). Phytochemical Characterization and Antifungal Activity of Marchantia polymorpha L. Against Rhizoctonia solani. Pharmacological Research - Modern Chinese Medicine, 11. https://doi.org/10.1016/j.prmcm.2024.100426
Piechota, G. (2021). Siloxanes in biogas: Approaches of Sampling Procedure and GC-MS Method Determination. Molecules, 26(7). https://doi.org/10.3390/molecules26071953
Poveda, J. (2024). Analysis of Marchantia polymorpha–microorganism interactions: Basis for Understanding Plant–Microbe and Plant–Pathogen Interactions. Frontiers in Plant Science, 15, 1–15. https://doi.org/10.3389/fpls.2024.1301816
Rafi, S. M., Patan, A., & Chandra, Y. P. (2025). A Review of SwissADME in Morden Medicinal Chemistry : Insights on Anti-Cancer, Anti-Tubercular, and Anti-Microbial Agents. International Journal of Current Trends in Pharmaceutical Research, 13, 90–96. https://doi.org/10.30904/j.ijctpr.2025.4865
Rahman, A. B. Z. N., Patwary, M. F., Ahmed, S., Hossen, M., Hossain, M. H., Islam, M. S., Rahman, R., Dutta, S., Sultana, K. F., Hossain, M. S., Ahmed, F., & Dhar, S. B. (2025). Antidermatophytic activity of Curcuma longa against Trichophyton spp.: Compound Identification and Molecular Docking to Lanosterol 14α-demethylase. BMC Complementary Medicine and Therapies, 25(1). https://doi.org/10.1186/s12906-025-05049-9
Rauf, A., Khan, H., Khan, M., Abusharha, A., Serdaroğlu, G., & Daglia, M. (2023). In Silico, SwissADME, and DFT Studies of Newly Synthesized Oxindole Derivatives Followed by Antioxidant Studies. Journal of Chemistry, 2023. https://doi.org/10.1155/2023/5553913
Riyadi, P. H., Romadhon, Sari, I. D., Kurniasih, R. A., Agustini, T. W., Swastawati, F., Herawati, V. E., & Tanod, W. A. (2021). SwissADME Predictions of Pharmacokinetics and Drug-Likeness Properties of Small Molecules Present in Spirulina platensis. IOP Conference Series: Earth and Environmental Science, 890(1). https://doi.org/10.1088/1755-1315/890/1/012021
Rocha, R. B., Alves, E. A., Tadeu, H. C., Teixeira, A. L., Bizzo, H. R., Antoniassi, R., Pacheco, S., & Santiago, M. C. P. de A. (2023). Caffeine and Chlorogenic Acid Content of Coffea canephora Cultivars in Different Environments. Coffee Science, 18. https://doi.org/10.25186/.v18i.2164
Sardar, H. (2023). Drug like potential of Daidzein using SwissADME prediction: In silico Approaches. PN: Phytonutrients, 2, 2–8.
Sauerschnig, C., Doppler, M., Bueschl, C., & Schuhmacher, R. (2018). Methanol generates Numerous Artifacts During Sample Extraction and Storage Of Extracts in Metabolomics Research. Metabolites, 8(1), 1–19. https://doi.org/10.3390/metabo8010001
Setyati, D., Su’udi, M., Ravitamala, E. S., Miladina, F. F., Babudin, B., Utarti, E., Arimurti, S., Nugraha, A. S., Putri, Y. A., Farhan, A. M., & Ulum, F. B. (2024). Antimicrobial and Phytochemistry study of Dendrobium linearifolium Teijsm. & Binn. from Gumitir, Jember, Indonesia. BIO Web of Conferences, 101, 1–12. https://doi.org/10.1051/bioconf/202410101001
Shah, M., Zia, M., Ahmad, I., Khan, M. U., Ejaz, H., Alam, M., Aziz, S., Nishan, U., Dib, H., Ullah, R., & Ojha, S. C. (2025). Computational analysis of Ayurvedic Metabolites for Potential treatment of Drug-resistant Candida auris. Frontiers in Cellular and Infection Microbiology, March. https://doi.org/10.3389/fcimb.2025.1537872
Singh, S., Bhadauriya, G., Agrawal, A., Bhati, V., Gore, S., & Rathor, K. S. (2024). Cytotoxicity and Antioxidant Activities of Methanolic Extract of Marchantia polymorpha and Dicranum scoparium using Network Pharmacology, molecular docking, and experimental approaches. Indian Journal of Natural Products and Resources, 15(4), 500–514. https://doi.org/10.56042/ijnpr.v15i4.10026
Singh, S., Gore, S., Gupta, S., & Singh, G. K. (2023). Natural Extracts from Marchantia polymorpha against Plant Pathogens Growth Inhibition. Environment Conservation Journal, 24(2), 311–319. https://doi.org/10.36953/ECJ.23562618
Son, N. C. T., Tan, T. Q., Lien, D. T. M., Huong, N. T. M., Tuyen, P. N. K., Phung, N. K. P., Phuong, Q. N. D., & Thu, N. T. H. (2020). Five phenolic compounds from Marchantia polymorpha L. and Their in Vitro antibacterial, Antioxidant and Cytotoxic Activities. Vietnam Journal of Chemistry, 58(6), 810–814. https://doi.org/10.1002/vjch.202000088
Soriano, G., Kneeshaw, S., Jimenez-Aleman, G., Zamarreño, Á. M., Franco-Zorrilla, J. M., Rey-Stolle, M. F., Barbas, C., García-Mina, J. M., & Solano, R. (2022). An Evolutionarily Ancient Fatty Acid Desaturase is Required for the Synthesis of Hexadecatrienoic Acid, Which Is The Main Source of the Bioactive Jasmonate in Marchantia polymorpha. New Phytologist, 233(3), 1401–1413. https://doi.org/10.1111/nph.17850
Spinedi, N., Storb, R., Aranda, E., Romani, F., Svriz, M., Varela, S. A., Moreno, J. E., Fracchia, S., Cabrera, J., Batista-García, R. A., de León, I. P., & Scervino, J. M. (2021). Ros-scavenging Enzymes as an Antioxidant Response to High Concentration of Anthracene in the Liverwort Marchantia polymorpha L. Plants, 10(7). https://doi.org/10.3390/plants10071478
Stelmasiewicz, M., Światek˛, Ł., & Ludwiczuk, A. (2022). Phytochemical Profile and Anticancer Potential of Endophytic Microorganisms from Liverwort species, Marchantia polymorpha L. Molecules, 27(1). https://doi.org/10.3390/molecules27010153
Stelmasiewicz, M., Świątek, Ł., & Ludwiczuk, A. (2023). Chemical and Biological Studies of Endophytes Isolated from Marchantia polymorpha. Molecules, 28(5). https://doi.org/10.3390/molecules28052202
Sumardi, & Suprianto. (2024). Assessing Drug-Likeness The Natural Compounds of Polar Extract Curcuma xanthorrhiza Rhizome via Lipinski ’ s Rules with SWISSADME Web Tool. Jurnal Indah Sains Dan Klinis, 05(03), 15–18. https://doi.org/10.52622/jisk.v5i3.03
Takemura, M., Hamada, T., Kida, H., & Ohyama, K. (2012). Cold-induced Accumulation of ω-3 Polyunsaturated fatty acid in a Liverwort, Marchantia polymorpha L. Bioscience, Biotechnology and Biochemistry, 76(4), 785–790. https://doi.org/10.1271/bbb.110915
Takizawa, R., Hatada, M., Moriwaki, Y., Abe, S., Yamashita, Y., Arimitsu, R., Yamato, K. T., Nishihama, R., Kohchi, T., Koeduka, T., Chen, F., & Matsui, K. (2021). Fungal-Type Terpene Synthases in Marchantia polymorpha are Involved in Sesquiterpene Biosynthesis in Oil Body Cells. Plant and Cell Physiology, 62(3), 528–537. https://doi.org/10.1093/pcp/pcaa175
Tamaian, R., Ciucure, C., & Gean, E. (2023). Comprehensive Investigation of Antimicrobial and Antifungal Mechanistic Pathways of Bioactive Phytochemicals from Apple Pomace Using Molecular Docking. Biology and Life Sciences Forum, 26(1). https://doi.org/10.3390/Foods2023-15034
Tran, T. Q., Phan, H. N., Bui, A. L., & Quach, P. N. D. (2020). Biological activities of in vitro liverwort Marchantia polymorpha L. extracts. Notulae Botanicae Horti Agrobotanici Cluj-Napoca, 48(2), 826–838. https://doi.org/10.15835/nbha48211884
Triviño, F. J. N., Velasco Amador, J. P., & Rivera Ruiz, I. (2025). Seborrheic Dermatitis Revisited: Pathophysiology, Diagnosis, and Emerging Therapies—A Narrative Review. Biomedicines, 13(10), 1–17. https://doi.org/10.3390/biomedicines13102458
Upadhyay, D. B., Mokariya, J. A., Nogales, J., Patel, S. G., Das, A., Nandi, A., More, N., Kumar, A., Rajani, D. P., Narayan, M., Kumar, J., Banerjee, S., Sahoo, S. K., & Patel, H. M. (2024). Indole clubbed 2,4‐thiazolidinedione linked 1,2,3‐triazole as a Potent Antimalarial and Antibacterial Agent Against Drug‐Resistant Strain and Molecular Modeling Studies. Arch Pharm Chemistry in Life Science, November 2023. https://doi.org/10.1002/ardp.202300673
Verpoorte, R., Kim, H. K., & Choi, Y. H. (2022). Trivialities in Metabolomics: Artifacts in Extraction and Analysis. Frontiers in Molecular Biosciences, 9, 1–11. https://doi.org/10.3389/fmolb.2022.972190
Warfield, J., Setzer, W. N., & Ogungbe, I. V. (2014). Interactions of Antiparasitic Sterols with Sterol 14α-demethylase (CYP51) of Human Pathogens. SpringerPlus, 3(1), 1–11. https://doi.org/10.1186/2193-1801-3-679
Yergaliyeva, E. M., Bazhykova, K. B., & Langer, P. (2022). In silico drug-likeness , Biological Activity and Toxicity Prediction of new 3, 5-bis (hydroxymethyl ) tetrahydro- 4H-pyran-4-one. Chemical Bulletin of Kazakh National University, 4, 14–20. https://doi.org/10.15328/cb1272
Zhang, J. Z., Wang, C., Zhu, T. T., Fu, J., Tan, H., Zhang, C. M., Cheng, A. X., & Lou, H. X. (2022). Spatial Distribution, Antioxidant Capacity, and Spore Germination-Promoting Effect of Bibenzyls from Marchantia polymorpha. Antioxidants, 11(11). https://doi.org/10.3390/antiox11112157
Zubair, M. S., Anam, S., Khumaidi, A., Susanto, Y., Hidayat, M., & Ridhay, A. (2023). Molecular Docking Approach to Identify Potential Anticancer Compounds from Begonia (AIP Conference Proceedings). https://doi.org/10.1063/1.4958513.
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Bioeksperimen: Jurnal Penelitian Biologi

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.










