Morphological and genetic identifications of Spirulina based on 16S rRNA gene
DOI:
https://doi.org/10.23917/bioeksperimen.v12i1.14879Keywords:
Arthrospira, phylogenetic, morphology, Molecular docking, spirulinaAbstract
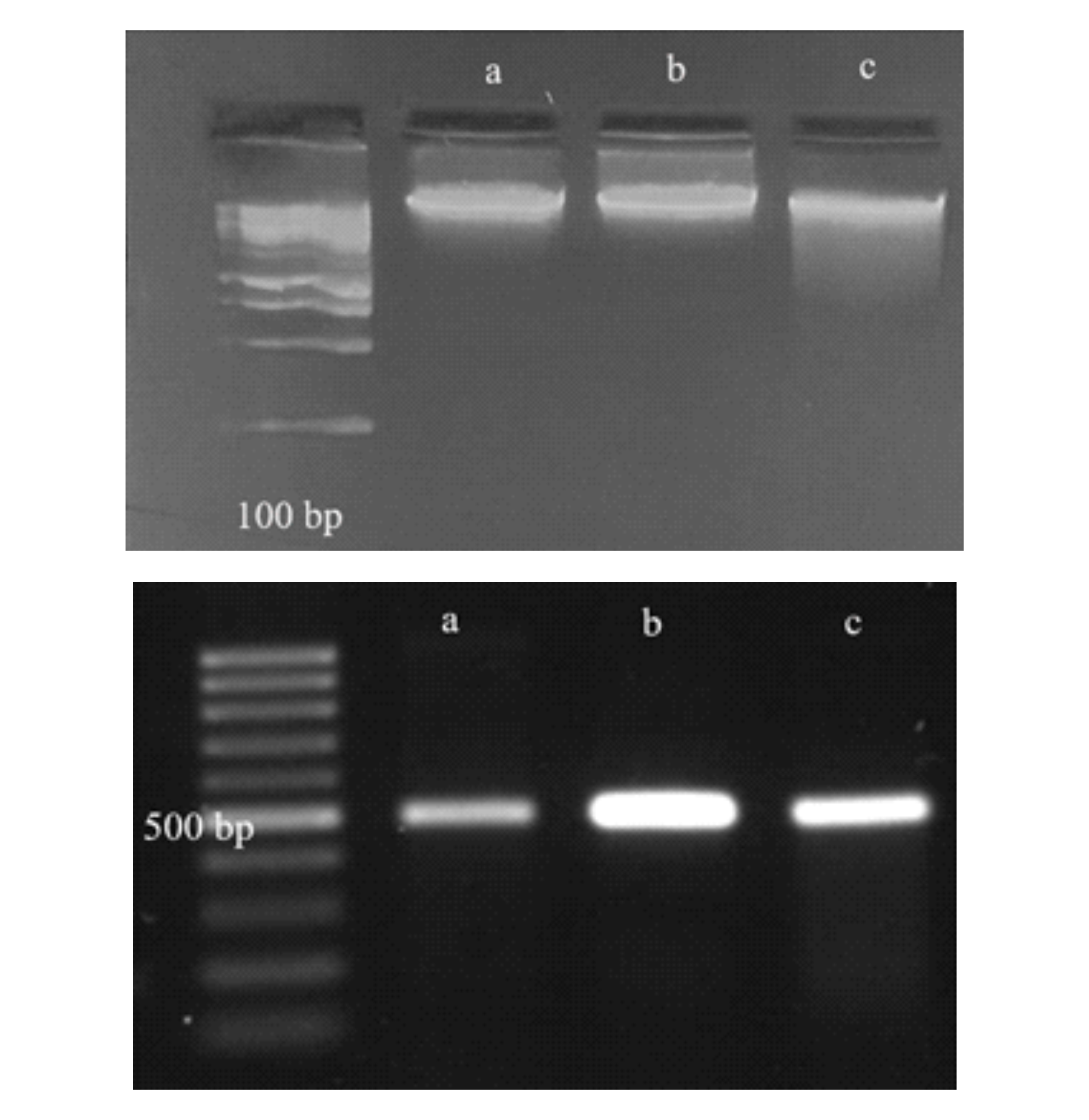
Spirulina is a microalga with spiral or linear trichome shapes that has potential in industrial and food sectors due to its rich nutritional content. Spirulina can live in various aquatic habitats with different environmental conditions. Differences in cultivation environments can affect the morphological characteristics of Arthrospira due to phenotypic plasticity, making morphology-based identification prone to errors. Therefore, molecular identification using the 16S rRNA gene for phylogenetic analysis is necessary. This study aims to identify and analyze the genetic relationships of Spirulina from freshwater and brackish water based on 16S rRNA gene sequences. Freshwater Spirulina samples were taken from Bogor (BGR) and Yogyakarta (MRP), while brackish water Spirulina was taken from Jepara (JPR). DNA extraction was performed using the Zymo Research Quick-DNA Fungal/Bacterial Miniprep Kit, amplification of the 16S rRNA gene was conducted through PCR, followed by DNA sequencing. Phylogenetic analysis was carried out using MEGA11 to construct the phylogenetic tree. The morphology of strains BGR and MRP showed straight and thin filaments, whereas JPR had spiral and relatively long filaments. Sequencing analysis results showed that strains JPR and MRP had the closest homology similarity to Arthrospira sp. IAQUASC-C0001 with percentages of 99.34% and 99.12%, respectively. Meanwhile, Spirulina from BGR had the closest homology similarity to Arthrospira fusiformis strain AICB 668 with a percentage of 99.78%. Phylogenetic analysis indicated that strains BGR, JPR, and MRP are more closely related to Arthrospira than to Limnospira.
Downloads
References
Alfaruqi, H. Q. D. (2021). Kajian Molekuler Pada Probiotik Asal Air Susu Ibu Dalam sintesis eksopolisakarida (EPS). Jurnal Bioteknologi dan Biosains Indonesia, 8(1), 114-123. 10.29122/jbbi.v8i1.4554
Aristya, G. R., Judith, T. P., Kasiamdari, R. S., Damaiyani, J., & Arif, M. F. (2025). Molecular identification and phylogenetic reconstruction of sugarcane ('Saccharum officinarum'L.) cultivars from Indonesia based on'rbc'L chloroplast gene. Australian Journal of Crop Science, 19(6), 620-632. https://search.informit.org/doi/10.3316/informit.T2025072900005300939774002
Balouch, H., Zayadan, B.K., Sadvakasova, A.K., Kossalbayev, B.D., Bolatkhan, K., Gencer, D., Civelek, D., Demirbag, Z., Alharby, H.F. and Allakhverdiev, S.I., 2023. Prospecting the biofuel potential of new microalgae isolates. International Journal of Hydrogen Energy, 48(50), pp.19060-19073. https://doi.org/10.1016/j.ijhydene.2023.02.028
Buddington, K.K., J.B. Donahoo, and R.K., Buddington. 2002. Dietary oligofructose and inulin protect mice from enteric and systemic pathogens and tumor inducers. J. Nutr. 132:472-477. https://doi.org/10.1093/jn/132.3.472
Buwono, N. R., & Nurhasanah, R. Q. (2018). Studi Pertumbuhan Populasi Spirulina sp. pada Skala Kultur yang Berbeda [Study of Spirulina sp. Population Growth in The Different Culture Scale]. Jurnal Ilmiah Perikanan dan Kelautan, 10(1), 26-33. https://doi.org/10.20473/jipk.v10i1.8516
Chotchindakun, K., Buddhasiri, S., & Kuntanawat, P. (2024). Enhanced Growth and Productivity of Arthrospira platensis H53 in a Nature-like Alkalophilic Environment and Its Implementation in Sustainable Arthrospira Cultivation. Sustainability, 16(19), 8627. https://doi.org/10.3390/su16198627
Emam, H. A., Abdel-Sattar, E., Salama, M. M., Salem, M. A., & Hashem, M. M. (2025). Spirulina platensis: Unveiling phenotypic plasticity impact on its metabolic profile and bioactivity via chemometric analysis. Food Bioscience, 63, 105605. https://doi.org/10.1016/j.fbio.2024.105605
Furmaniak, M. A., Misztak, A. E., Franczuk, M. D., Wilmotte, A., Waleron, M., & Waleron, K. F. (2017). Edible cyanobacterial genus Arthrospira: Actual state of the art in cultivation methods, genetics, and application in medicine. Frontiers in Microbiology, 8, 2541. https://doi.org/10.3389/fmicb.2017.02541
Iqbal, M., Buwono, I. D., & Kurniawati, N. (2016). Analisis perbandingan metode isolasi DNA untuk deteksi White Spot Syndrome Virus (WSSV) pada udang Vaname (Litopenaeus vannamei). Jurnal Perikanan Kelautan, 7(1). https://jurnal.unpad.ac.id/jpk/article/view/13941
Johnson, M. S., Burns, B. P., Herdean, A., Angeloski, A., Ralph, P., Morris, T., ... & Larkum, A. W. (2022). A cyanobacteria enriched layer of Shark Bay stromatolites reveals a new Acaryochloris strain living in near infrared light. Microorganisms, 10(5), 1035. https://doi.org/10.3390/microorganisms10051035
Jung, C. H., Braune, S., Waldeck, P., Küpper, J. H., Petrick, I., & Jung, F. (2021). Morphology and growth of Arthrospira platensis during cultivation in a flat-type bioreactor. Life, 11(6), 536. https://doi.org/10.3390/life11060536
Karamalis, C., Xirogianni, A., Simantirakis, S., Delegkou, M., Papandreou, A., & Tzanakaki, G. (2025). Molecular Identification of Meningitis/Septicemia Due to Streptococcus spp. in Greece (2015–2024). Diagnostics, 15(13), 1632. https://doi.org/10.3390/diagnostics15131632
Ko, J. T., Li, Y. Y., Chen, P. Y., Liu, P. Y., & Ho, M. Y. (2024). Use of 16S rRNA gene sequences to identify cyanobacteria that can grow in far‐red light. Molecular Ecology Resources, 24(1), e13871. https://doi.org/10.1111/1755-0998.13871
Łach, Ł., Khomutovska, N., Kwiatowski, J., & Jasser, I. (2024). Testing 16S Primers for Proper Identification of Cyanobacterial Communities in Small Water Bodies. Water, 16(10), 1357. https://doi.org/10.3390/w16101357
Lao, I. K. M., & Edullantes, B. (2025). Growth, Productivity, and Size Structure of Spirulina Strain Under Different Salinity Levels: Implications for Cultivation Optimization. Phycology, 5(3), 31. https://doi.org/10.3390/phycology5030031
Lestari, M. D., & Pangastuti, A. (2025). Laju Pertumbuhan, Biomassa, dan Produksi Fikosianin Arthrospira platensis Perairan Pulau Jawa Pada Variasi Salinitas. Bioscientist: Jurnal Ilmiah Biologi, 13(4), 2848-2857. https://doi.org/10.33394/bioscientist.v13i4.18387
Li, M. N., Han, Q., Wang, N., Wang, T., You, X. M., Zhang, S., ... & Zhang, G. F. (2024). 16S rRNA gene sequencing for bacterial identification and infectious disease diagnosis. Biochemical and Biophysical Research Communications, 739, 150974. https://doi.org/10.1016/j.bbrc.2024.150974
Lijassi, I., Arahou, F., Mansouri, Z., Wahby, A., Rhazi, L., & Wahby, I. (2024). Comparative Analysis of Effect of Culture Conditions on Growth and C-Phycocyanin Production in Helical and Linear Spirulina. Current Microbiology, 81(6). https://doi.org/10.1007/s00284-024-03684-y
Lin, J. N., Lai, C. H., Yang, C. H., & Huang, Y. H. (2023). Validation of 16S rRNA and complete rpob gene sequence analysis for the identification of Elizabethkingia species. International Journal of Molecular Sciences, 24(16), 13007. https://doi.org/10.3390/ijms241613007
Mishra, P., Mishra, R. R., Prasad, S. M., & Nath, G. (2022). Isolation and molecular characterization of nutritionally potent Arthrospira maxima from Indian paddy field. Biocatalysis and Agricultural Biotechnology, 42, 102338. https://doi.org/10.1016/j.bcab.2022.102338
Misztak, A. E., Waleron, M., Furmaniak, M., Waleron, M. M., Bazhenova, O., Daroch, M., & Waleron, K. F. (2021). Comparative genomics and physiological investigation of a new Arthrospira/Limnospira strain O9. 13F isolated from an alkaline, winter freezing, Siberian lake. Cells, 10(12), 3411. https://doi.org/10.3390/cells10123411
Nosratimovafagh, A., Esmaeili Fereidouni, A., & Krujatz, F. (2023). Effect of light spectrum, salinity, and glucose levels on Spirulina morphology. Journal of the World Aquaculture Society, 54(6), 1687-1701. https://doi.org/10.1111/jwas.13012
Puspitasari, A., Budiarsa, I. M., Ashari, A., Dhafir, F., Windarsih, Y., & Trianto, M. (2025). Analisis Filogenetik Cryptic species Apis cerana Fabricius, 1793 Berdasarkan Gen 16S rRNA. Bioscientist: Jurnal Ilmiah Biologi, 13(1), 521-530. https://doi.org/10.33394/bioscientist.v13i1.14967
Rilisa, C & Suantika,G. (2021). Biological, Technical, and Financial Feasibilities Study Spirulina sp. Biomass Production with Modified Commercial Medium in Indonesia, 3BIO: Journal of Biological Science, Technology and Management, 3(2):67-76. https://doi.org/10.5614/3bio.2021.3.2.2
Selvendran, D. (2015). Large scale algal biomass (Spirulina) production in India. In Algal biorefinery: An integrated approach (pp. 151-167). Cham: Springer International Publishing. https://doi.org/10.1007/978-3-319-22813-6_7
Sinetova, M. A., Kupriyanova, E. V., & Los, D. A. (2024). Spirulina/Arthrospira/Limnospira—Three names of the single organism. Foods, 13(17), 2762. https://doi.org/10.3390/foods13172762
Stover, N. A., & Cavalcanti, A. R. (2017). Using NCBI BLAST. Current Protocols Essential Laboratory Techniques, 14(1), 11-1. https://doi.org/10.1002/cpet.8
Supriatna, M., Mahmudi, M., & Musa, M. (2020). Model pH dan hubungannya dengan parameter kualitas air pada tambak intensif udang vaname (Litopenaeus vannamei) di Banyuwangi Jawa Timur. JFMR (Journal of Fisheries and Marine Research), 4(3), 368-374. https://doi.org/10.21776/ub.jfmr.2020.004.03.8
Susintowati, S. H., Puniawati, N., Poedjirahajoe, E., & Handayani, N. S. N. (2018, July). Plastisitas Fenotip Gastropoda Mangrove Taman Nasional Alas Purwo: Morfometri Cangkang Dan Operkulum. In PROSIDING SEMINAR NASIONAL. https://ejurnal.unisri.ac.id/index.php/prosemnas/article/view/2105
Szubert, K., Toruńska-Sitarz, A., Stoń-Egiert, J., Wiglusz, M. and Mazur-Marzec, H., 2021. Comparative characterization of two cyanobacteria strains of the order Spirulinales isolated from the Baltic Sea-polyphasic approach in practice. Algal Research, 55, p.102170. https://doi.org/10.1016/j.algal.2020.102170
Thevarajah, B., Nishshanka, G. K. S. H., Premaratne, M., Nimarshana, P. H. V., Nagarajan, D., Chang, J. S., & Ariyadasa, T. U. (2022). Large-scale production of Spirulina-based proteins and c-phycocyanin: A biorefinery approach. Biochemical Engineering Journal, 185, 108541. https://doi.org/10.1016/j.bej.2022.108541
Ujwala, et al. (2025). Life between Salt and Fresh: The Unique World of Brackish Water Habitats. Journal of Marine Science: Research & Development, 15(1), pp. 490. https:// www.omicsonline.org/open-access-pdfs/life-between-salt-and-fresh-the-uniqueworld-of-brackish-water-habitats.pdf
Wan, D., Wu, Q., & Kuča, K. (2016). Spirulina. In Nutraceuticals (pp. 569-583). Academic Press.
Zapata, D., Arroyave, C., Cardona, L., Aristizábal, A., Poschenrieder, C., & Llugany, M. (2021). Phytohormone production and morphology of Spirulina platensis grown in dairy wastewaters. Algal Research, 59, 102469. https://doi.org/10.1016/j.algal.2021.102469
Zhang, J., Xue, H., Yan, S., Shi, H., Du, L., Zhang, J., ... & Ma, C. (2022). Phylogenetic analysis of Arthrospira strains from Ordos based on 16S rRNA. Scientific Reports, 12(1), 14375. https://doi.org/10.1038/s41598-022-18695-7.
Submitted
Accepted
Published
How to Cite
Issue
Section
License
Copyright (c) 2026 Bioeksperimen: Jurnal Penelitian Biologi

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.










